How chronic inflammation results in DNA damage and hypomethylation associated with cancer initiation
A new study led by researchers at Queen Mary University of London describes how chronic inflammation leads to altered, tumour-like epigenetic state and the re-expression of ancient, gene-editing retrotransposons that induce DNA damage.
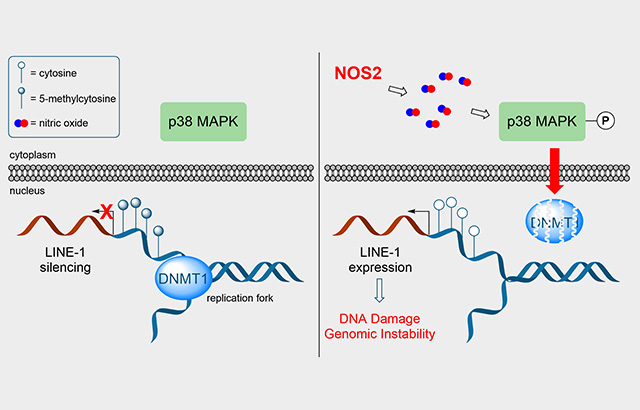
NOS2 promotes passive DNA demethylation and LINE-1 expression. (Left) Under normal physiological conditions, DNMT1 methylates the nascent DNA strand during replication and LINE-1 retrotransposons are silenced by promoter CpG methylation. (Right) NOS2 expression results in sustained p38-MAPK activation, which leads to DNMT1 protein degradation. Replication in the absence of DNMT1 activity results in passive DNA demethylation, LINE-1 expression, and genomic instability.
In this Q&A, study lead Dr Christopher Switzer talks about why this is not only a consequence of nitric oxide signalling, which is linked to cancer formation, but also of other cellular stressors that cause cancer. More broadly, he explains why the mechanism described may be the common cause of most human cancers and may provide clues on other the genesis of other inflammatory diseases.
What is new about the study?
Inducible nitric oxide synthase (NOS2), an inflammation-associated enzyme, is associated with poor cancer patient outcomes in breast and brain cancers, yet the molecular mechanisms linking NOS2 activity with cellular transformation and cancer progression are not clear. In our study, we show that NOS2 alters the epigenetic state of human cells by decreasing DNA methylation. Loss of DNA methylation is a common early feature of human cancers, yet the cause of this phenomenon is not known. Here we identify a molecular mechanism of how NOS2 activity leads to decreased DNA methylation that occur early on in cancer formation.
Is there anything surprising about the results?
We thought that NO would directly inhibit DNMT1 catalytic function and that the effect would be specific to NO. Surprisingly, the loss of DNMT1 and DNA hypomethylation required sustained p38 MAPK activity, which indicates that other chronic activators of p38, such as the known oncogenic EBV, mutant Ras and cytokine signalling, may also induce DNA damage and DNA hypomethylation via this mechanism.
Why is the study important?
Inflammation has long been associated with cancer initiation and progression. For the first time, the molecular links between chronic inflammation and the early steps of epigenetic and cellular reprogramming have been described. This is a major break-through, as other long-lasting diseases such as arthritis and diabetes involve inflammation signals coupled to cellular reprogramming. Therefore, the mechanism we describe will have further implication for conditions chronic inflammation. Perhaps more important than the altered DNA methylation and cellular reprograming, the loss of genomic methylation activates ancient viral elements that go on to damage the genome, another molecular event observed early in cancer formation. Therefore, we link the epigenetic destabilization of cells with chronic inflammation.
What are the wider implications?
While we demonstrate that NOS2 induces DNA methylation changes via cellular stress mechanisms, we also indicate that this is a generic feature of cellular stress. This is important, as other inducers of human cancer also activate the same stress pathways as NOS2. Therefore, we think we have identified a universal mechanism that results in cellular transformation.
Additionally, based on this work, we are following up our study with related projects in diabetes and radiation-induced cellular transformation and cardiac fibrosis. Therefore, the broader consequences are that inflammatory mechanisms of cellular reprogramming have been identified for further exploration, which we hope will lead to novel anti-inflammatory drugs.
Further information
Research paper: ‘NOS2 and S-nitrosothiol signaling induces DNA hypomethylation and LINE-1 retrotransposon expression’, Switzer CH, Cho HJ, Eykyn TR, Eaton P, PNAS.
Download PDF [PDF 2,009KB]