FAQs
Is the study end date going to be extended?
The recruitment period in the study has been extended to 31st December 2025 and the new study end date is 31st December 2026.
Are you recruiting new sites into the study?
Yes, we are taking on new sites in the UK Adult ITP Registry. If you are interested in joining, please email us the following information (uk-itp.registryteam@nhs.net):
- Name of the NHS organisation taking part in the study.
- Name of hospital/site where participants will be consented.
- Name of Principal Investigator at site. Post/Job Title of PI; email and telephone number.
- Predicted participant recruitment at your site (please state numbers per month/year)
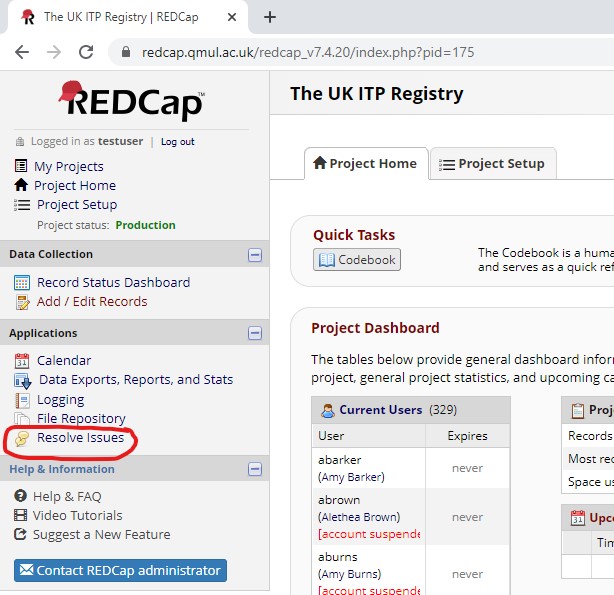
Where can I access all the queries on REDCap for our site?
You will be able to see all the queries by clicking on the ‘Resolve Issues’ link once you’ve logged into REDCap, under the ‘Applications’ section on the left hand side of the screen:
For any questions where the data is missing or unavailable, please enter, ‘9999’ for numeric fields and ‘01/01/1000’ for date fields. Examples can be found in our REDCap user guides. Please ensure that you have the source documents from the patients medical notes or electronic records to confirm any data being entered.
What is the process for recruiting patients?
Each site will be provided with a site-specific starter pack once all approvals have been obtained. When a patient has consented to the study, please send us the consent forms with the updated 'Participants log', via our email, uk-itp.registryteam@nhs.net. You will have to also upload your recruitment activity on your Local Portfolio Management System (LPMS) in order for your activity to be linked to the NIHR Central Portfolio Management System. Please check with your R&D team to confirm what LPMS your trust uses (most of our sites use EDGE but other systems include ReDa, StudyLine, R-Peak and Documas). At the end of every month, we will confirm the research activity for each site, cross checking the data on CPMS with the participants logs and consent forms we have received from you.
The participants log is an Excel Spreadsheet which keeps a record of every patient who has consented to the registry and also gives the unique 'Registry Identification Code' (RIC) in order to enter data on the online database anonymously. This is not a screening log and should only contain a list of patients who have consented to the study. Sites can then log onto the online database (REDCap) and register the patients and enter the information that is required in the registry for each patient using their respective RICs.
The REDCap database can be accessed via https://redcap.qmul.ac.uk/.
The initial/baseline data (data from the date of ITP diagnosis)should be collected within 6 weeks of a patient consenting to the study. The baseline data consists of the following forms: Participant Details; Clinician Information; Anthropometric and Lifestyle Data; Bleeding Events; Splenectomy; ITP Treatments; Supportive Therapies, Comorbidities; Family History; Biochemical Tests; Haematological Tests; Immunological Tests; Coagulation Fields; Full Blood Counts (Hbs, Neutrophils, plts); Bone Marrow Biopsy; and DAT Indium Scanning.
Once the initial data entry is complete for the baseline data, patients must be followed-up at least once annually for data entry onto REDCap (this includes completion of the following forms Bleeding events; ITP treatments; Supportive therapies; Comorbidities; and Full Blood Counts (Hbs, Neutrophils, plts). All the other forms would have been completed during initial data entry but can be updated if necessary at follow up.
With the registry study, the more data we have the better our analysis. Of course with primary ITP, patients may attend clinics multiple times a year and we appreciate that this would be too burdensome for research staff to constantly update every visit. Therefore, we request sites to follow patients and update information on each patient at least once annually at the very minimum.
Can patients exclusively be consented to the pregnancy arm of the registry?
No, if you have any patients who have a confirmed diagnosis of ITP prior to or during their pregnancies, they must first be consented, using the relevant consent forms, and registered to 'Arm 1: Primary ITP Registry' (for Primary ITP patients) or 'Arm 3: Secondary ITP Registry' (for Secondary ITP patients) and then the pregnancy registry arm ('Arm 2: Pregnancy Registry').
If a patient has been prescribed a medication where the dose is being tapered, are we required to capture every single dose change?
Ideally we would like each dose change captured on REDCap, however, we are aware that this is not always possible for all patients and so we have added the option to select tapering of doses on REDCap now which you can select (but please ensure you have provided the treatment start and end date).
Is there any funding or support for sending blood samples?
Unfortunately, we do not have funding to support reimbursement of postage and supplies for the blood sample collection aspect of the protocol. Only sites that have the capacity to do so should collect blood samples.
Is it compulsory to use the proformas/worksheets for initial and follow-up data entry?
No, these documents have been created to aid your data entry; you do not have to use them if you do not wish to do so.
Do the REDCap user guides and paper proformas/worksheets have separate approval?
The user guides and paper proformas do not require separate approval for use. These are intended to help sites to enter data on the database for an existing protocol that already has approval. We recently made changes to the online REDCap database to help streamline and make data entry more efficient for sites; these documents go hand in hand with the new database.
How long the documents should be archived for?
QMUL policy is 20 years. As we are not requesting for physical data to be sent to us, sites should follow their local archiving policy.
Is recruitment for the UKITPR halted in light of COVID-19?
Patient safety is paramount and we understand if sites decide to halt recruitment for the UKITPR in line with their local policies and use clinical staff aligned to research projects to support NHS resource demands. You do not have to inform us that this is the case; please proceed as instructed by your local research management teams.